Leucidal® Liquid
Background
Over the past several decades there has been growing public pressure, increasingly strict chemical regulations, preservative sensitization issues, and the potential for developing microbial resistance to the chemical preservative products typically used in cosmetic and personal care formulations. These factors have resulted in numerous preservation chemicals being pulled from the marketplace, despite being the products of choice at one time. To offer a solution to this preservation paradigm, Active Micro Technologies (AMT) has developed a line of products based on naturally occurring compounds that provide active cosmetic properties, but by their very nature are also capable of providing product preservation. This antimicrobial capability is due to natural mechanisms developed by plants and microorganisms by which they protect themselves from their environment and other competing organisms.
Science
Leucidal® Liquid is based on an antimicrobial peptide originally derived from the lactic acid bacteria, Leuconostoc kimchii. L. kimchii is one of 15 species of microorganisms that make up the mixed culture responsible for producing the Korean dietary staple known as kimchi, a type of fermented cabbage.
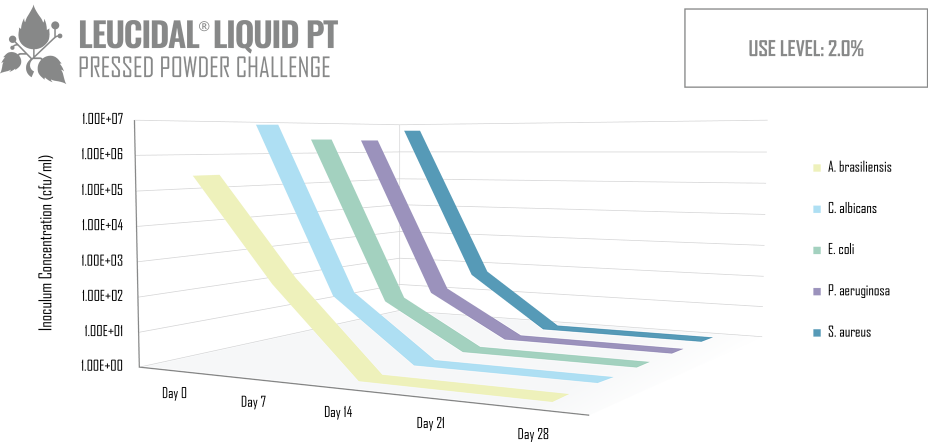
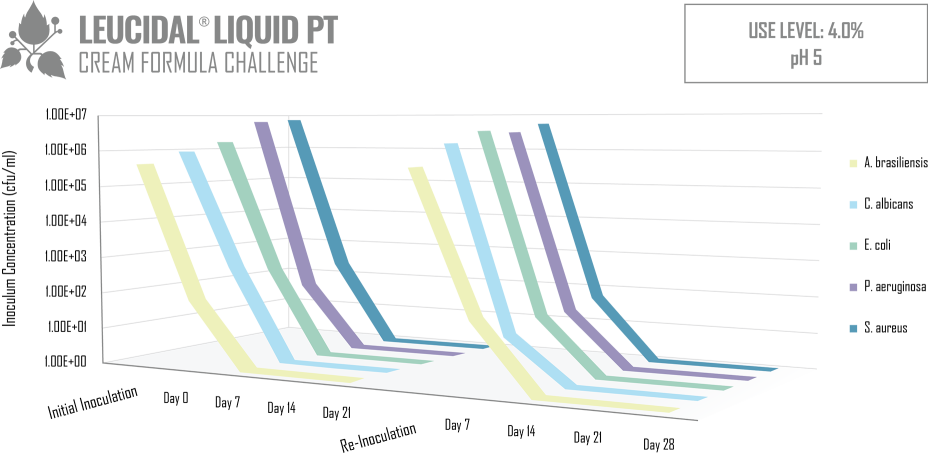
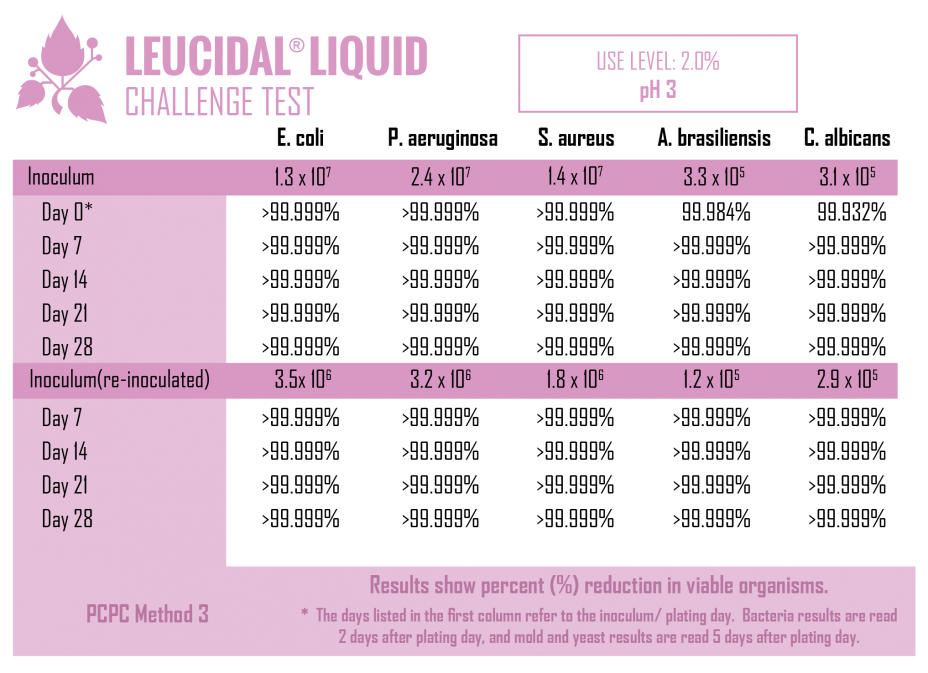
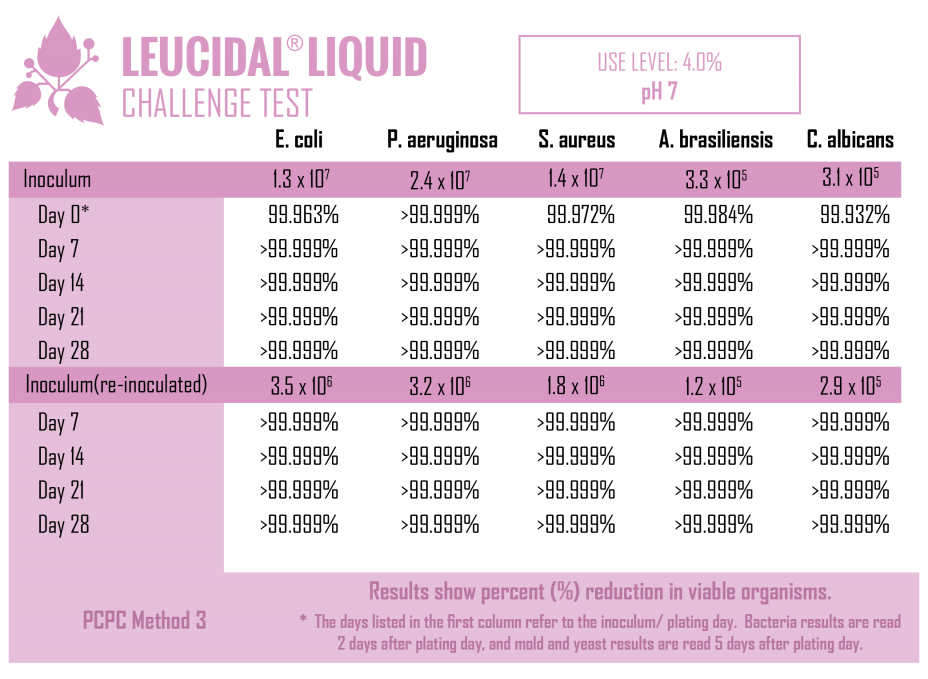
Like many lactic acid bacteria, L. kimchii is capable of restricting the growth of other microorganisms by acidifying its environment, but as is common in nature, it is not content to limit itself to a single mechanism of defense. In addition to acidifying its environment, it also produces a novel antimicrobial peptide. Using modern fermentation and bioprocessing technology, AMT has commercialized this antimicrobial peptide to produce Leucidal® Liquid. The first step in the development of this product was to determine the peptide’s potential ability to inhibit the growth of a variety of bacteria and fungi. Using standard serial dilution protocols in growth media, the Minimum Inhibitory Concentrations (MICs) for Leucidal® Liquid were determined for a variety of both bacterial and fungal organisms. The positive MIC screening results warranted further testing to confirm its ability to provide product preservation. A Double Challenge Test was completed using 2% Leucidal® Liquid in a generic cream base formulation. Samples were inoculated with E. coli, P. aeruginosa, S. aureus, K. pneumoniae, C. albicans, A. niger, and B. cepacia. During the first 28 day incubation period, samples were periodically collected and tested for the presence of these microorganisms. Following this initial 28 days of incubation, the cream samples were then re-inoculated with the microbial cultures and sampled over an additional 28-day period.
 Benefits
Benefits
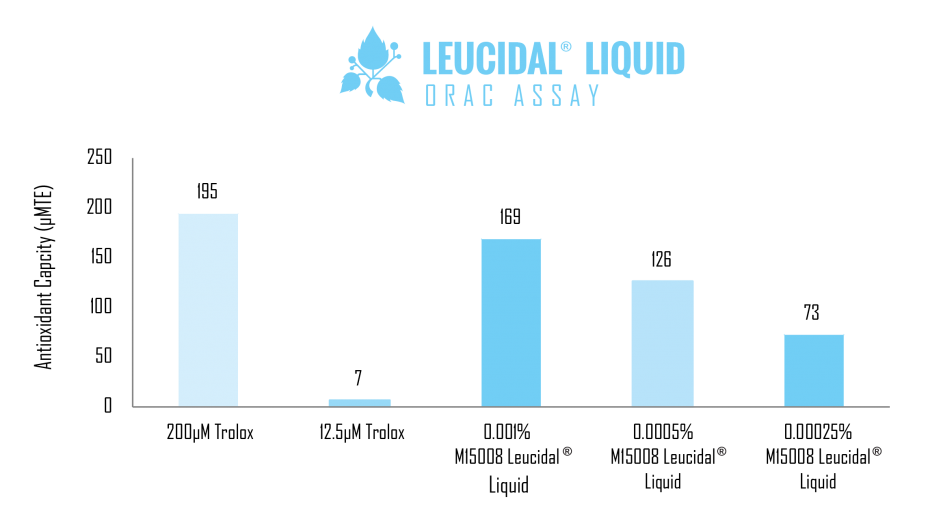
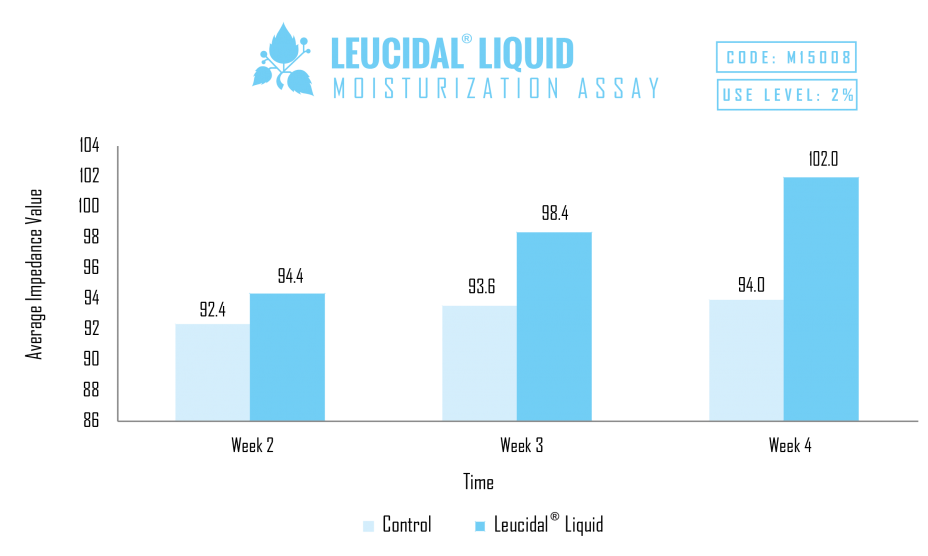
Because peptides have been found to provide skin moisturization properties, Leucidal® Liquid was used in a comparative study to evaluate its ability to provide cosmetic benefits, in addition to the demonstrated antimicrobial function. A skin moisturization study was performed using a generic cream base, compared with the same cream base containing 1% Leucidal® Liquid. As demonstrated by the results of this study, the addition of 1% Leucidal® Liquid to the base cream formulation provided a 10% increase in moisturization. Based on these results, adding this innovative product provides the formulator the opportunity to capitalize on both the natural antimicrobial properties of Leucidal® Liquid, as well as its ability to provide potent moisturizing benefits to the cosmetic formulation. These properties make it ideal for applications addressing numerous skin and scalp conditions.
Use Recommendations
As with all biological materials, some attention must paid to the conditions under which Leucidal® Liquid is used. Based on bench-scale evaluations, as well as actual product applications, Leucidal® Liquid has been found to be effective over a wide range of typical cosmetic and personal care product manufacturing conditions. The product has been found to be heat stable up to 70°C and active under both acidic (pH 3) and basic conditions (pH 8).